Conservation Journal
April 1994 Issue 11
The ageing & stabilisation of shellac varnish resin - an undergraduate research project at Imperial College
Having graduated from Imperial College's Chemistry department in 1992, and muddling through Kenya and Madagascar on a student expedition for a few months, I have eventually found myself among the chosen few of the RCA/V&A Conservation Course students.
The third year of my degree course gave the opportunity for a literature review project and a practical research project, together forming a major part of the year's work. I chose to relate these to conservation, as this seemed to be an area in which I could have a useful and enjoyable vocation, and the projects would give me an opportunity to find out more about it.
My literature review was entitled 'The Ageing and Stabilisation of Picture Varnish Resins'. This discussed some of the recent advances in the use of resins in picture varnishes, including the use of various stabilisers to prolong the useful life of the varnishes, the development of new synthetic resins, and the associated studies on the ageing processes and characteristics of the materials used. Emphasis was made on the chemical nature of the resins, their deterioration on ageing, and the stabilisers and new resins being used to produce improved varnishes. This review introduced me to the range of conservation literature available and also to some of the people in institutions in London, through contact with my external supervisors Alan Cummings at the RCA, and Raymond White at the National Gallery.
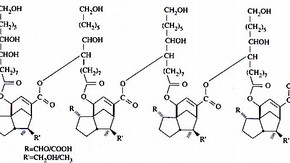
Research in new resins and methods of stabilising them is of importance in paintings conservation, but is rarely applied to furniture or wood coatings. Shellac is the most widely used natural resin applied in furniture making, restoration and conservation, but its properties make it less suitable for other uses. I thought that it would be worth applying some of the techniques used in picture varnish research to assess the stability of shellac, and to see if pale, stable varnishes could be prepared. These would be useful in certain circumstances where shellac had been used originally, but where the present colour must not be altered, for example, delicate shades of wood in marquetry or Tunbridgeware.
The purpose of my research project was to assess some of the chemical and physical changes that take place in shellac resin varnish films under prolonged accelerated light ageing, and to determine what effects if any, that modern hindered amine light stabilisers (HALS) have upon the deterioration of shellac films. Some thermal ageing experiments were also carried out and experiments were made to decolourise various grades of shellac using activated charcoal.
The light ageing experiments were carried out using shellac films on glass microscope slides, placed in a Microscal light-fastness tester, illuminated at 50,000 lux for a total of 2031 hours. Several grades of shellac were tested, including both bleached shellacs and dewaxed flake shellacs. Two similar HALS additives were used, Tinuvin 123 and 292, in 0.5% and 1.0% concentrations. Sample sets were also kept at 80 lux, in the dark, and in an oven at 50°C. The amount of light received was also referenced with blue wool standards. Samples of shellac were scraped from the slides at regular intervals for analysis and observation during the experiment. The results of the experiment showed that bleached shellac films are not very stable at high light intensities, becoming bloomed, yellowed, brittle and blistered. Dewaxed flake shellac films were more stable, particularly the palest grades, although they became fairly brittle and some colour changes and cracking were observed. The instability of bleached shellac is mainly due to changes induced by the harsh manufacturing process, which leaves a residual 0.1-0.2% bound chlorine in the shellac. Bleached shellac is unstable in storage and commercially is not intended for long term use. It should not be used on metals, or for any conservation purpose.
The use of Tinuvin 123 and 292 hindered amine light stabilisers seemed to have a definite effect on reducing the effects of degradation especially in bleached shellac, with Tinuvin 123 being the most effective. However, more comprehensive studies would have to be made to determine whether these additives were of any real value, and whether or not there were any Side effects. After 18 months, solutions of shellac containing the stabilisers appear slightly more yellow than those without.
It seemed apparent that a major cause of deterioration in shellac is due to heating processes undergone during its manufacture into flake shellac, when temperatures of over 70°C can be encountered. However, some heating is necessary to make the shellac hard enough, and varnishes made from seedlac, the crude material which has had little heat treatment, does not form very hard films, and damages easily when applied to a wooden surface.
The experiments to decolourise shellac were quite profitable, although the process is time consuming and messy. The palest commercial grade of flake shellac is called Platina, a dewaxed decolourised grade, and is a lot more coloured than bleached shellac. After stirring a solution with activated charcoal for some time, and filtering under vacuum, the resultant solution is slightly paler than that of bleached shellac, being a pale yellow colour. The films obtained from these solutions are virtually colourless. These experiments showed that shellac varnishes could be made as pale as bleached shellac, by using commercial grades of flake shellac and activated charcoal, with a minimum of equipment.
Shellac varnishes can go off if stored for a long time due to esterification between acid groups in the shellac constituent compounds and the alcohol solvent. This is more apparent in the palest grades and can result in the varnish not drying properly.
Overall shellac varnish films are quite stable, and evidence from objects polished with shellac shows that it lasts for a long time, although it is susceptible to damage from abrasion, alcohol or water. It should remain easily removable with ethanol, and can be considered a reversible treatment where the object will not be affected by a polar solvent such as ethanol.
This project proved very interesting and enjoyable and helped to encourage my interest in conservation as a career, and I am very grateful to those people who helped with it. Copies of my reports are available if anyone is interested.
April 1994 Issue 11
- Editorial
- A saint unveiled
- 17th Century Chinese canopied bed
- Recording the changes: V&A conservation departmental archive
- The ageing & stabilisation of shellac varnish resin - an undergraduate research project at Imperial College
- Treatment of an early 18th Century Indian chintz qanat
- ICCROM - Japanese paper conservation course 1993: an introduction to the ancient skills of scroll mounting for the modern conservator
- Sand cast aluminium