Conservation Journal
Spring 2007 Issue 55
Investigation of the patina on Bidri using advanced surface analysis techniques

Figure 1. The Bidri box used for this experimental study Photography by Emma Beardmore (click image for larger version)
The name given to the Indian metalwork, Bidri, is derived from the city of Bidar, where it has been produced since at least the seventeenth century. It is composed of an alloy of zinc (~95%) and copper (~5%) and is heavily inlaid or overlaid with silver and possibly gold or brass to produce an intricate design before it is finally polished. The metalwork is unusual in that the Bidri alloy is patinated, forming a deep black surface that contrasts with the various metal inlays (Figure 1). Although there have been a few studies of Bidri1,2,3, there has, as yet, been no full characterisation of the patina.
As an initial indication of whether Bidri could indeed be a predominately zinc based alloy, the densities of two Bidri objects, acquired solely for the purpose of research, were determined. This simple test is useful as an initial screening procedure. Calculating their densities involved submerging the two objects in water for a short period of time. One of the test items, the Bidri disc, exhibited a white corrosion product on the surface as a result of this short exposure to water. However, the surface of the Bidri box inlayed with a white metal exhibited no notable difference in appearance although it had had the same exposure time to water. This highlighted a difference between the compositions of the two objects and/or a difference in the surface treatment; for example perhaps a waxy polish had been applied to the box, but not to the disc. The results of the density measurements found the Bidri box and disc to have mean densities of 6.98g cm-3 and 6.77g cm-3 respectively, which are comparable to zinc's density of 7.14g cm-3, and sit within a 10% error margin.
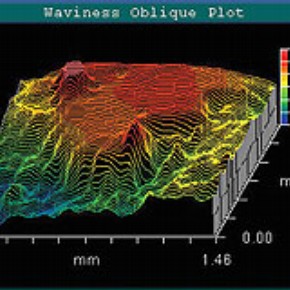
Mapping the surface of the Bidri box using a Zygo white-light optical interferometer enabled the roughness of the patina to be seen. As shown in Figure 2, the patina is somewhat uneven, which is expected, considering that the Bidri is handmade. The difference in height between the white metal inlay and the matt black surface of the patina varied, however; the black patina tended to be about 2.5µm below the inlay. This technique also revealed small, neat, circular indents in the surface of the inlay. It was concluded that this could be where the metal had been hammered into the engravings.
EDXRF (energy dispersive X-ray fluorescence) is a non-destructive, non-intrusive technique that can be used to find the elemental composition of a sample. A Röhntec ArtTax EDXRF was used with an air path and a 30W molybdenum target X-ray tube. This procedure was employed on the two Bidri objects and the spectra produced revealed that they were both alloys of zinc and copper, and that the white metal which had been inlayed on the surface of the Bidri box was silver. More unexpectedly, it was found that the Bidri box contained quantities of nickel and the disc showed small traces of arsenic on the patina.
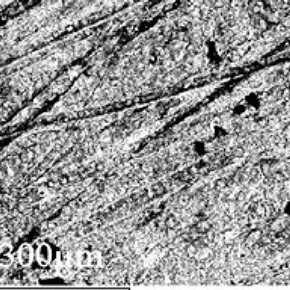
Figure 3. A backscattered electron image, produced at a magnification of 200 X (click image for larger version)
In order to manipulate the quantitative data produced by EDXRF spectrometry, zinc and copper alloy standards were examined using this technique to produce calibration curves. The curves show the relationship between the abundance of an element (percent) in a sample, and the number of times it was detected using the EDXRF. This allowed an estimation to be made about the percentage composition of zinc and copper in the Bidri samples - without any sample surface preparation. It was found that the Bidri box was approximately 85% zinc and 4% copper, whilst the Bidri disc was in the magnitude of 90% zinc and 3% copper, which are near the expected values. It would appear that the EDXRF underestimates the abundance of zinc in the samples but does confirm that zinc is the major component and copper a significant trace element. More work is required to make the EDXRF more accurate.
A small disc was cut from the underside of the Bidri box to be viewed at high magnification using the scanning electron microscope (SEM). The general structure found was as expected for a metal alloy, however many linear features, about five microns in width, were observed on the surface of the patina (Figure 3). Operating the electron microscope enabled a technique called X-ray mapping to be used. This approach shows the elemental composition of a sample in more detail, showing the regions where each element is present at high concentrations. This technique confirmed again that the Bidri was a zinc and copper alloy, containing traces of silicon, carbon, oxygen and chlorine, but the imaging also revealed calcium traces (Figure 4) which were found in the linear features in much higher abundance than elsewhere on the patina suggesting a calcium rich deposit had been laid down during polishing or through scratching.
Figure 4. A backscattered electron image, produced at a magnification of 200 X, showing the abundance of calcium (shaded white) in the linear features (click image for larger version)

Figure 5. A Secondary Ion (SIM) image of the ion beam milled trench, viewed at a 45° tilt, showing the patinated layer on top of the metal. (click image for larger version)
The SIMS technique removes atoms from the surface, so by recording the elemental composition of the sample as a function of time, the variation of elements with depth can be found. It took three scans before the depth profile detected any elements, suggesting there may be some type of polish or layer of wax protecting the top of the patina. The depth profile also revealed that the proportions of zinc and copper increased with depth, and elements like calcium and sodium decreased with depth. This indicates that the calcium and sodium are surface contaminants.
These initial experiments have been very useful but it is clear that the characterisation of Bidri is by no means complete. Several attempts were made at locating interested individuals with workshop experience with Bidri; however this yielded no successful results. A number of experimental advances have been made gathering more information about the patina of the Bidri as a material. However, there is still a mystery surrounding the deep black colour of the patina and the compound causing it. The project described here has demonstrated the potential of a variety of surface analysis techniques and further studies are planned during the period 2006/7 to further unravel the mystery of Bidri patina.
Acknowledgments
The authors are grateful to Magnus Lekstrom and David Collins for their contributions to the analyses and to Susan Stronge and Susan La Niece for their helpful advice and for donating the samples of Bidri used in this study.
References
1. Craddock, Paul. 'Enigmas of Bidri', Surface Engineering, 2005, Vol. 21, pp. 5-6.
2. Stronge, S. Bidri Ware: Inlaid Metalwork from India, (London, 1985).
3. La Niece, S. and Martin, G. 'The Technical Examination of Bidri Ware', Studies in Conservation, (1987), Vol. 32, pp. 97-101.
Spring 2007 Issue 55
- Editorial Comment - Conservation Journal 55
- Stein Mellon Textile Project at the V&A
- Storage of the Stein Loan Collection
- Discoveries on a pair of cassoni
- Treatment of two Coptic tapestry fragments
- Surreal semi-synthetics
- Investigation of the patina on Bidri using advanced surface analysis techniques
- Printer friendly version